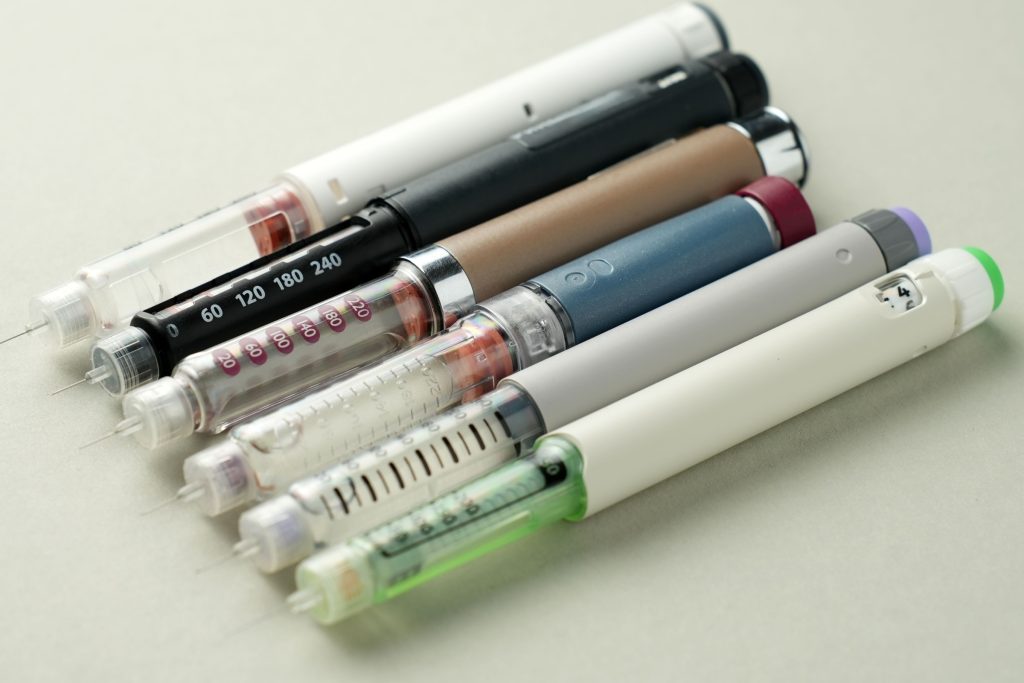
The latest weight-loss treatment shift is being driven by pills, not just injections. Oral obesity drugs are expanding the market because they offer a simpler option for people who do not want weekly shots. Reuters and other reports say Novo Nordisk’s oral Wegovy has already entered the U.S. market, while Eli Lilly’s oral candidate orforglipron remains under FDA review.
That convenience matters. Doctors say some patients hesitate to start injectable GLP-1 drugs even when they are interested in treatment. A pill can lower that barrier, especially in primary care, where many obesity prescriptions now begin. This appears to be one reason oral products are drawing so much attention.
There is also a practical difference between the two-pill approaches. Reports say Lilly’s orforglipron may be easier to take because it does not have the same strict timing and fasting requirements associated with oral semaglutide. That could make adherence simpler in real life.
Newer Drugs Are Raising the Ceiling
The second part of this weight-loss treatment shift involves stronger next-generation drugs. The biggest name in that group is retatrutide, Eli Lilly’s experimental “triple-G” medicine, which targets GLP-1, GIP, and glucagon pathways. Reuters reported on March 19 that in a late-stage diabetes trial, retatrutide reduced A1C by 1.7% to 2.0% and produced up to 16.8% average weight loss at 40 weeks.
Earlier studies had already raised expectations. Reporting on prior retatrutide data showed weight loss approaching 29% in some obesity-related trials, which is well above what many current drugs achieve. That has made the drug one of the most closely watched candidates in the field.
Novo Nordisk is also trying to move higher. The FDA approved a higher-dose version of Wegovy, called Wegovy HD, on March 19, 2026. Reuters said the new 7.2 mg version delivered an average weight loss of about 20.7% over 72 weeks in clinical testing, with a U.S. launch planned for April.
Taken together, these developments show the market splitting into at least two tracks. One track focuses on access and convenience through pills. The other focuses on stronger weight loss through more potent injectable combinations.
Cost and Coverage Are Becoming Just as Important
This weight-loss treatment shift is not only about science. It is also about who can afford these drugs and how they get them. Recent reporting says the oral products helped push entry-level out-of-pocket prices lower through pricing deals tied to federal policy, even as insurance coverage remains inconsistent.
That inconsistency may matter more than the list price. In many cases, patients still face major gaps in coverage for obesity drugs, especially when insurers treat them as optional rather than essential. Some health plans have already cut or narrowed coverage due to cost pressures, leaving patients to pay out of pocket or switch products. This is an inference drawn from the market reporting and current payer behavior.
For patients, that means cost and convenience may shape decisions as much as medical performance. A person may choose a pill because it is easier to take, easier to get, or cheaper out of pocket, even if another drug produces more weight loss in trials.
More Power Brings More Questions
Doctors also warn that more effective drugs can bring new risks. Reuters reported that retatrutide caused more gastrointestinal side effects than some current options, including nausea and vomiting. Other coverage has also pointed to unusual sensations, such as skin tingling in earlier studies.
There is also concern about inappropriate use. More powerful weight-loss drugs may help people with severe obesity or those who did not respond enough to current therapy. But physicians have warned that the same potency could create problems if people use them for minimal or cosmetic weight loss. That concern has followed the GLP-1 market for years and may grow as stronger drugs enter the market.
The field is now moving faster than it was just a few years ago. Oral options are expanding. Stronger injectables are closing in. Prices are shifting, but access still depends heavily on coverage. That makes this more than a product cycle. It is a broader weight-loss treatment shift that could reshape who uses obesity medicine, how they use it, and what doctors consider realistic results.